Treatment of Knee Joint Pain: The 3-Pain-Mechanism Map That Explains Why the Same Diagnosis Gets Completely Different Therapies
Treatment of Knee Joint Pain: The 3-Pain-Mechanism Map That Explains Why the Same Diagnosis Gets Completely Different Therapies
Introduction: Why the Same Diagnosis Produces Completely Different Treatment Plans
Two patients sit in separate examination rooms. Both have received the same diagnosis: knee osteoarthritis. Both report moderate pain that disrupts daily activities. Yet when they leave, one carries a prescription for hydrotherapy sessions and a knee brace, while the other receives a corticosteroid injection and a nerve-targeting medication. Same diagnosis, radically different therapies. The question patients rarely think to ask is: why?
The answer lies in a fundamental truth that transforms how knee joint pain should be understood and treated. Knee osteoarthritis is not a single biological event. It arises from at least three distinct pain mechanisms—inflammatory, mechanical, and neuropathic—and treatment efficacy depends entirely on which mechanism dominates in a given patient.
The scale of this problem demands attention. According to the GBD Study 2021, 374.7 million people worldwide suffered from knee osteoarthritis as of 2021. The Lancet Rheumatology (2023) projects a 74.9% increase in knee OA cases by 2050. In the United States alone, osteoarthritis generates an estimated $65 billion annually in direct healthcare costs.
Yet the healthcare system is failing these patients. According to research published in ScienceDaily (March 2026), more than 60% of OA patients receive treatments that clinical guidelines do not recommend. Fewer than half are referred to exercise programs or physical therapy. Approximately 40% are sent to a surgeon before non-surgical options have been properly explored.
This article provides the mechanistic education that most patient resources lack. It maps every major treatment tier to the specific pain pathway it targets, explains why acetaminophen fails while hydrotherapy succeeds for certain patients, and examines what the emerging pipeline—including the historic January 2026 lorecivivint NDA submission—means for the future of knee OA care.
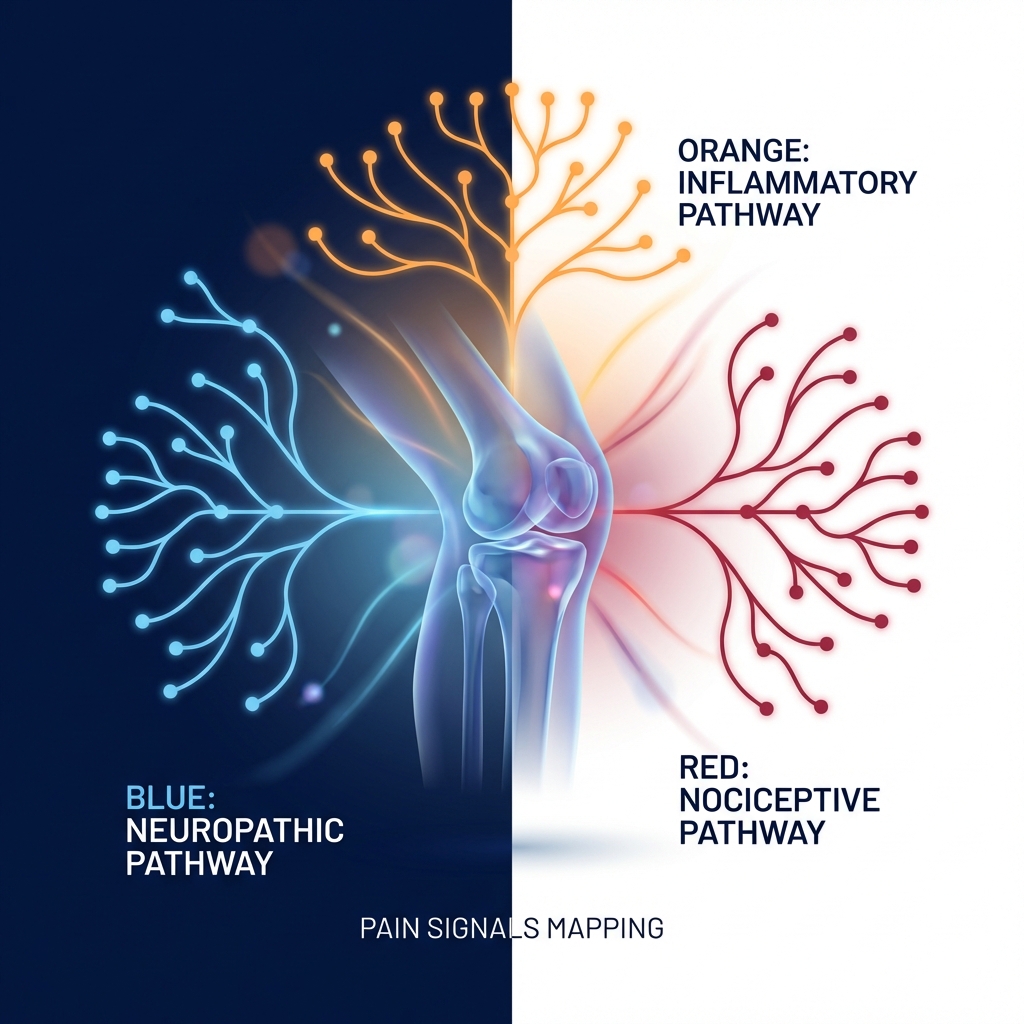
The 3-Pain-Mechanism Map: A Framework for Understanding Knee OA Treatment
Pain in knee osteoarthritis is not monolithic. It originates from three biologically distinct pathways, each requiring a different therapeutic approach. According to research published in Frontiers in Medicine (2025), understanding these mechanisms is essential for tailored management.
The three mechanisms form a conceptual map:
- Inflammatory pathway — driven by synovitis, cytokines, and immune activation
- Mechanical pathway — driven by cartilage degradation, altered joint biomechanics, and structural load distribution
- Neuropathic pathway — driven by central sensitization, peripheral nerve sensitization, and altered pain processing
Most patients with knee OA have overlapping contributions from all three pathways. However, the dominant mechanism varies by individual, disease stage, and phenotype. This explains why subtype-aware treatment is essential—and why the same diagnosis produces completely different therapies.
Mechanism 1: The Inflammatory Pathway — When the Joint Becomes Its Own Enemy
The inflammatory pathway begins when the synovial membrane becomes inflamed (synovitis), triggering the release of pro-inflammatory cytokines including TNF-α, IL-1β, and IL-6. These molecules activate matrix metalloproteinases that degrade cartilage and amplify pain signaling. Abnormal new blood vessel growth (neovascularization) in the synovium perpetuates this inflammation cycle.
Inflammatory pain presents clinically as morning stiffness, warmth, swelling, pain at rest, and pain that responds to anti-inflammatory agents. Patients with a dominant inflammatory phenotype require treatments that specifically target this cascade.
High BMI represents the single most critical modifiable risk factor for knee OA and directly amplifies the inflammatory pathway. Adipose tissue produces pro-inflammatory adipokines. According to Frontiers in Public Health (2025), a 5-unit BMI increase in women corresponds to a 35% increased risk of developing knee OA.
Treatments That Target the Inflammatory Pathway
Topical NSAIDs serve as first-line pharmacological therapy. Topical diclofenac and ketoprofen inhibit COX-1 and COX-2 enzymes locally, reducing prostaglandin synthesis in the synovium without the systemic gastrointestinal, cardiovascular, and renal risks of oral NSAIDs. OARSI and ACR guidelines strongly recommend topical NSAIDs as first-line pharmacotherapy.
Oral NSAIDs provide the same COX inhibition at a systemic level but carry significantly higher risk profiles—particularly cardiovascular risk with selective COX-2 inhibitors and gastrointestinal risk with non-selective agents.
Why acetaminophen fails represents a critical patient education point. Acetaminophen does not inhibit COX enzymes in peripheral inflamed tissue at therapeutic doses and has no meaningful anti-inflammatory action in the joint. A network meta-analysis showed it performs no better than placebo for OA pain—only a 4mm difference on a 0–100mm VAS scale. Despite this evidence, it remains one of the most commonly prescribed analgesics for knee OA.
Intra-articular corticosteroids suppress the inflammatory cascade by inhibiting NF-κB signaling, reducing cytokine production, and decreasing synovial fluid volume. They prove effective for acute inflammatory flares, though repeated injections may accelerate cartilage loss over time.
Hyaluronic acid injections restore the viscoelastic properties of synovial fluid, reduce friction, and may have direct anti-inflammatory effects by binding to CD44 receptors on synoviocytes. High-molecular-weight hyaluronic acid shows particular benefit in the inflammatory subtype.
Hydrotherapy outperforms land-based exercise for inflammatory subtypes because water’s buoyancy reduces joint loading forces by up to 50–75%, allowing therapeutic movement without mechanically stressing an inflamed joint. Water’s hydrostatic pressure also reduces synovial swelling. The 2025 PLOS One network meta-analysis of 139 RCTs covering 9,644 patients ranked hydrotherapy second overall among non-drug therapies and first specifically for pain relief.
Genicular artery embolization (GAE) represents a minimally invasive option that selectively embolizes the abnormal neovascular supply to the inflamed synovium, reducing the blood flow that sustains chronic synovitis.
Mechanism 2: The Mechanical Pathway — When Structure Drives Pain
The mechanical pathway activates when cartilage degradation reduces the joint’s ability to distribute load evenly, creating focal stress concentrations on subchondral bone. Bone remodeling, osteophyte formation, and meniscal pathology alter joint kinematics, generating pain through mechanoreceptors and nociceptors in bone, periosteum, ligaments, and joint capsule.
Mechanical pain worsens with activity and weight-bearing, improves with rest, and associates with crepitus, joint instability, and reduced range of motion. Cartilage itself has no nerve supply, so cartilage loss does not directly cause pain—pain arises from the secondary structural consequences on innervated tissues.
Treatments That Target the Mechanical Pathway
Knee braces redistribute compressive forces away from the most damaged compartment of the knee, reducing focal bone stress and pain. The 2025 PLOS One network meta-analysis ranked knee braces highest overall among 12 non-drug therapies for reducing pain, improving function, and easing stiffness.
Exercise therapy strengthens the quadriceps and surrounding musculature, improving dynamic joint stability and reducing peak joint loading during gait. However, a February 2026 meta-analysis raised controversy by suggesting exercise may offer only small and short-lived relief for OA—an important nuance that underscores the need for mechanism-targeted treatment selection.
Weight management produces mechanical benefits because each pound of body weight reduction decreases knee joint loading force by approximately four pounds during walking.
Total knee arthroplasty (TKA) represents the definitive mechanical intervention—replacing damaged joint surfaces eliminates the structural source of mechanical pain. Over 600,000 knee replacements are performed annually in the United States. However, studies suggest up to 80% of patients told they need TKA may not actually require surgery, underscoring the importance of exhausting non-surgical knee treatment options first.
Lorecivivint and the DMOAD pipeline target the mechanical pathway at the molecular level. In January 2026, Biosplice Therapeutics submitted an NDA to the FDA for lorecivivint, a CLK/DYRK kinase inhibitor. Its mechanism involves selective inhibition of DYRK1A (reducing inflammation and catabolic enzymes) and CLK2 (providing cartilage-protective properties) via Wnt pathway modulation—a first-in-class approach. Despite decades of research, no disease-modifying osteoarthritis drug (DMOAD) has yet been approved.
Mechanism 3: The Neuropathic Pathway — When the Nervous System Amplifies Pain
The neuropathic pathway activates when chronic joint injury and inflammation sensitize peripheral nociceptors (peripheral sensitization) and alter central pain processing in the spinal cord and brain (central sensitization). The result is pain disproportionate to the degree of structural damage visible on imaging.
Neuropathic pain presents as widespread pain beyond the knee, allodynia, hyperalgesia, poor correlation between imaging findings and pain severity, and inadequate response to anti-inflammatory or mechanical interventions.
Treatments That Target the Neuropathic Pathway
Duloxetine (SNRI) inhibits the reuptake of serotonin and norepinephrine in descending pain modulation pathways, reducing central sensitization. It is one of the few pharmacological agents with evidence specifically for centrally sensitized OA pain.
Radiofrequency ablation (RFA) of genicular nerves uses thermal energy to disrupt the genicular nerve branches that transmit pain signals from the knee to the brain. Mechanistically distinct from GAE, RFA targets the neuropathic pathway rather than inflammation.
Low-dose radiation therapy, long used in Germany and Spain, passed a rigorous placebo-controlled trial in October 2025, demonstrating real benefits for knee OA pain beyond placebo according to the American Society for Radiation Oncology. This treatment remains largely unknown in English-speaking countries.
Opioids are not recommended. Despite targeting pain centrally, oral and transdermal opioids are strongly contraindicated by ACR/Arthritis Foundation guidelines for knee OA due to risks of addiction, cognitive effects, and hyperalgesia with chronic use.
The Cellular and Regenerative Tier: Targeting Multiple Pathways Simultaneously
Cellular and regenerative therapies occupy a unique position because they can simultaneously address inflammatory, structural, and potentially neuropathic pathways through paracrine signaling, immunomodulation, and tissue regeneration.
PRP (Platelet-Rich Plasma) releases growth factors that modulate local inflammation, promote tissue repair signaling, and may stimulate chondrocyte proliferation. It primarily targets the inflammatory pathway with secondary structural effects.
BMAC (Bone Marrow Aspirate Concentrate) contains mesenchymal stem cells that exert potent immunomodulatory effects and can differentiate toward chondrogenic lineages, addressing both inflammatory and structural pathways.
Exosome therapy delivers regenerative signals through extracellular vesicles without the risks associated with live cell transplantation.
Stanford Medicine researchers (November 2025) discovered that blocking 15-PGDH—a “gerozyme” that increases with age—reversed cartilage loss in aging mouse knee joints and triggered early cartilage regeneration in human tissue from knee replacement surgeries. This approach works by reprogramming existing chondrocytes rather than transplanting stem cells.
The Treatment Access Gap: Why Mechanism Knowledge Matters
The systemic failure in OA care is stark: more than 60% of patients receive treatments that clinical guidelines do not recommend. When clinicians and patients do not understand which pain mechanism is dominant, they default to generic protocols—often prescribing acetaminophen, which has no meaningful mechanistic basis for OA pain, or escalating prematurely to surgery.
The 3-mechanism framework is not merely academic. It is the practical foundation for understanding why a treatment that works for one patient fails completely for another with the same diagnosis.
Conclusion: Mechanism Is the Map — Treatment Is the Territory
The reason the same diagnosis of knee OA produces completely different therapies is that “knee joint pain” is not a single disease. It is a clinical syndrome driven by inflammatory, mechanical, and neuropathic mechanisms in varying proportions in each patient.
Understanding the mechanism behind a treatment is not just academic knowledge—it is the foundation for asking better questions, making more informed decisions, and advocating for personalized care.
Explore Mechanism-Targeted Treatment for Knee Pain
Understanding the three mechanisms driving knee joint pain is the first step. The next step is identifying which mechanism—or combination of mechanisms—is dominant in a specific case.
Unicorn Bioscience develops treatment protocols based on individual patient factors including inflammation levels, age, injury type, current medications, and personal health goals. A multi-modal treatment menu—encompassing PRP, BMAC, exosome therapy, hyaluronic acid injections, and stem cell therapy—allows for mechanism-targeted approaches rather than one-size-fits-all protocols.
All injections are administered using ultrasound and X-ray guidance to ensure accurate delivery to the targeted treatment area. Virtual and in-person consultations are available across eight locations in Texas, Florida, and New York, with same-day treatment available for qualified candidates.
To discuss which treatment approach aligns with specific pain mechanisms and individual health goals, contact Unicorn Bioscience at (737) 347-0446 or visit unicornbioscience.com.
Schedule Your Consultation Today!