Cerebral Palsy Stem Cell Therapy: Experimental Treatments vs. Proven Orthopedic Solutions
Cerebral Palsy Stem Cell Therapy: Experimental Treatments vs. Proven Orthopedic Solutions
Families navigating a cerebral palsy diagnosis often find themselves searching for every possible avenue of treatment. The hope driving that search is both understandable and powerful. In 2026, stem cell therapy continues to generate significant interest as a potential breakthrough for neurological conditions, including cerebral palsy. Yet the critical reality remains unchanged: despite more than 15 years of research and over 2,400 children treated in clinical studies, no stem cell therapies for cerebral palsy have received FDA approval—or approval in any global jurisdiction.
This article provides families with a clear framework for understanding two distinct pathways: experimental neurological stem cell treatments that remain outside proven scope, and evidence-based orthopedic interventions for the musculoskeletal complications that frequently accompany cerebral palsy. The goal is to help families distinguish legitimate clinical research from stem cell tourism while accessing proven orthopedic care available today.
Understanding Cerebral Palsy: The Neurological vs. Musculoskeletal Divide
Cerebral palsy is a neurological disorder affecting movement and posture, impacting approximately 1 in 345 children in the United States. An estimated 764,000 Americans currently live with CP, making it the most common childhood motor disability. Global prevalence ranges from 1.6 per 1,000 live births in high-income countries to 3.4 per 1,000 in low and middle-income nations.
The primary neurological damage occurs in the developing brain—and current medical interventions cannot reverse this injury. However, cerebral palsy creates a cascade of secondary effects throughout the body. This distinction between the neurological cause (brain injury) and secondary musculoskeletal complications (contractures, hip dysplasia, scoliosis, joint deformities) is essential for understanding treatment options.
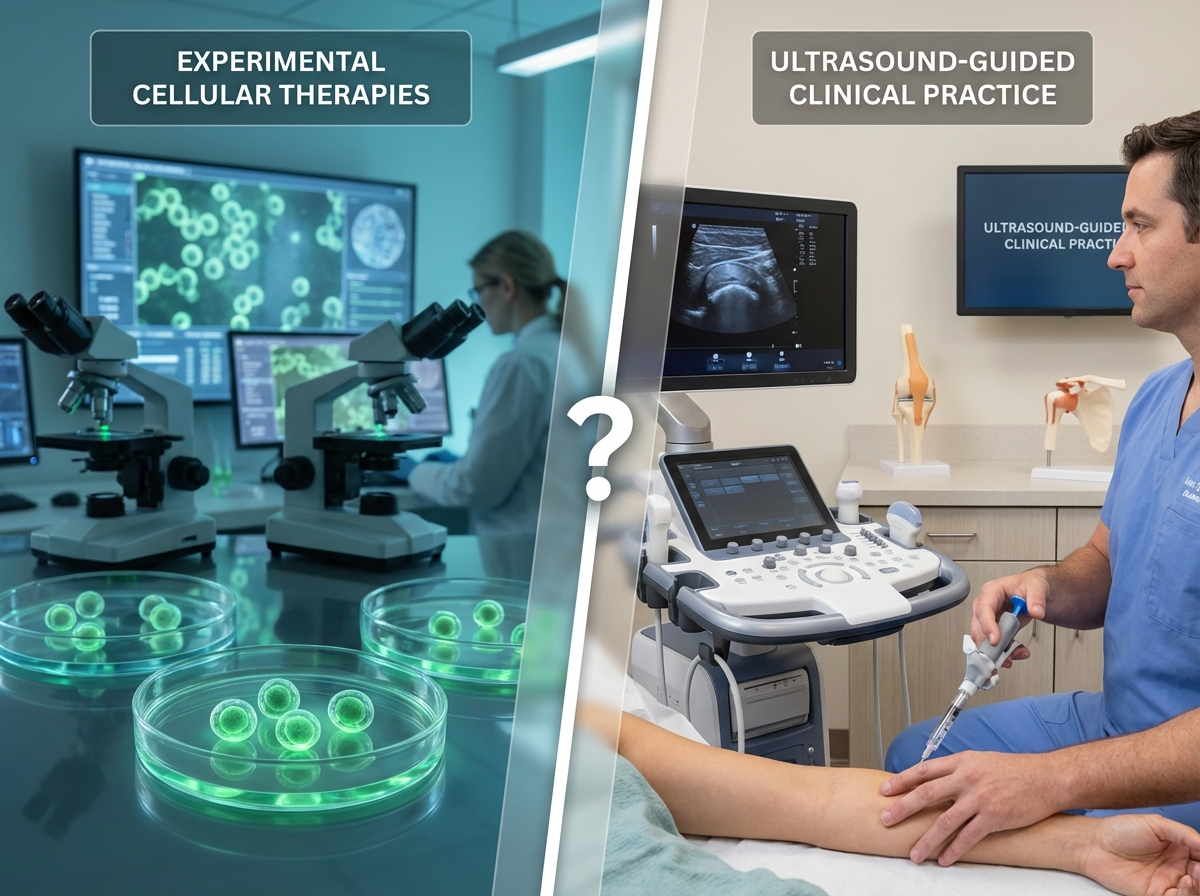
Stem cell therapy targets the neurological component, attempting to repair or regenerate neural tissue. Orthopedic interventions, by contrast, address the physical complications that develop over time. The fundamental reality families must understand: neurological repair remains experimental, while musculoskeletal management is evidence-based and available now.
The Current State of Stem Cell Research for Cerebral Palsy in 2026
The regulatory landscape for stem cell therapy in cerebral palsy remains unchanged from previous years—zero approved therapies exist despite extensive research since the early 2010s. A meta-analysis published in Pediatrics in April 2025 demonstrated that umbilical cord blood can improve gross motor function, though results remain heterogeneous across studies.
Researchers continue investigating multiple stem cell types:
- Umbilical cord blood (UCB) — currently showing the most promise
- Mesenchymal stem cells (MSCs) — derived from various tissues
- Bone marrow mononuclear cells
- Neural progenitor cells
Administration routes vary significantly across trials, including intravenous, intrathecal, and intramuscular delivery. According to research published in Neural Regeneration Research, 39 clinical trials were registered on clinicaltrials.gov as of 2020, with vast differences in protocols making direct comparison difficult.
Why Stem Cell Therapy Isn’t Approved Yet: The Scientific Barriers
The path to FDA approval faces multiple scientific obstacles. Current evidence suggests stem cells likely work through anti-inflammatory and immune-modulating effects rather than actual cell replacement. This paracrine signaling promotes endogenous repair but doesn’t regenerate damaged neural tissue as originally hoped.
Several barriers prevent regulatory approval:
- Heterogeneous trial designs with varying protocols
- Small sample sizes limiting statistical power
- Manufacturing challenges in producing consistent cellular products
- Quality control issues across different facilities
- Timeline complexity — benefits appear 3-6 months post-treatment, with full potential at 6-12 months
Notably, younger patients under five years show greater improvement in trials than older children, complicating the development of standardized treatment protocols. The ethical complexities of pediatric research further slow progress compared to adult applications.
Legitimate Research Pathways vs. Stem Cell Tourism
Families seeking stem cell treatment must distinguish between legitimate research and commercial exploitation. Duke University operates an FDA-registered expanded access program (NCT03327467) that launched in October 2017. Europe launched a similar expanded access program in Poland in 2019 through the FamiCord Group.
Stem cell tourism presents significant dangers. Families pay $10,000 to $60,000 or more for unproven treatments overseas without regulatory oversight. City of Hope and other medical institutions have issued warnings about these unregulated clinics.
Red flags for identifying stem cell tourism scams include:
- Guaranteed results or cure claims
- Lack of institutional review board oversight
- Treatments in countries with minimal regulation
- No published research or safety data
- Pressure to make quick decisions
Insurance typically does not cover stem cell therapy for CP because it remains experimental and is not considered standard of care.
The Financial and Physical Risks of Unproven Treatments
The financial burden of stem cell tourism is substantial. Families spend $10,000 to $60,000 or more for overseas treatments, while lifetime CP care costs already exceed $1.6 million in 2025 dollars. CDC data indicates medical costs for children with CP are 10 to 26 times higher than for children without the condition.
Physical risks of unregulated procedures include infection, immune reactions, and unknown long-term effects. The opportunity cost is equally significant—money spent on unproven treatments could fund evidence-based therapies, adaptive equipment, and quality-of-life improvements that deliver measurable benefits.
Cerebral Palsy’s Musculoskeletal Complications: What Orthopedics Can Treat
While neurological repair remains experimental, the musculoskeletal complications of cerebral palsy represent a proven orthopedic problem with evidence-based solutions. Research published in Acta Orthopaedica confirms that joint contractures affect 60% of adults with CP.
The progression follows a predictable pattern: spasticity leads to dynamic contractures that become fixed without early intervention. Johns Hopkins Medicine identifies common orthopedic complications including:
- Hip dysplasia
- Neuromuscular scoliosis
- Equinovarus foot
- Flatfoot
- Toe walking
- Muscle contractures
Lower limb contractures occur first in the foot for GMFCS levels I-II and in the knee for GMFCS levels III-V. These secondary physical problems require orthopedic management—not experimental neurological treatments.
Evidence-Based Orthopedic Interventions for CP Complications
The proven orthopedic toolkit for CP complications includes multiple interventions supported by decades of clinical evidence:
- Botulinum toxin injections for spasticity management
- Selective dorsal rhizotomy for reducing spasticity
- Single-event multilevel surgery (SEMLS) addressing multiple issues simultaneously
- Tendon lengthening procedures for contracture correction
- Physical therapy for preventing contracture progression
According to research in Frontiers in Neurology, a multiple-disciplinary, family-centered, evidence-based management approach yields the best outcomes. Early identification and intervention prevent fixed contractures and progressive deformities. These interventions are FDA-compliant, typically insurance-covered, and supported by extensive clinical evidence.
FDA-Compliant Regenerative Solutions for CP’s Orthopedic Complications
Unicorn Biosciences offers FDA-compliant regenerative therapies specifically targeting the musculoskeletal complications of cerebral palsy—not attempting neurological repair. This distinction is critical for families evaluating treatment options.
Available therapies for CP-related orthopedic issues include:
- Platelet-Rich Plasma (PRP) therapy — addressing tendon injuries and inflammation from spasticity-related joint stress
- Hyaluronic acid injections — managing joint arthritis in CP patients with early-onset osteoarthritis
- Bone Marrow Aspiration Concentrate (BMAC) — supporting healing in contracture-related joint deterioration
Each injection utilizes precision-guided technology with ultrasound or X-ray imaging, ensuring accurate treatment delivery even in patients with altered anatomy. These autologous therapies use the patient’s own biological materials, maintaining FDA compliance while addressing documented orthopedic complications.
When Regenerative Orthopedic Therapy Is Appropriate for CP Patients
Appropriate candidates for regenerative orthopedic therapy include CP patients with documented musculoskeletal complications such as contractures, joint deterioration, and spasticity-related orthopedic issues. The evaluation process at Unicorn Biosciences includes:
- Comprehensive imaging review (X-rays, MRIs, CT scans)
- Assessment of inflammation levels
- Consideration of age, injury type, and location
- Review of current medications and personal health goals
- GMFCS level and ambulatory status evaluation
Integration with the existing CP care team—neurologists, physical therapists, and developmental pediatricians—ensures coordinated treatment planning. When traditional orthopedic surgery remains the better option, honest assessment guides appropriate referrals.
What CP Families Should Focus On Now: The 2026 Action Plan
Rather than waiting for experimental treatments, families should prioritize proven interventions available today:
- Early physical therapy and contracture prevention programs
- Evidence-based orthopedic care for emerging complications
- Quality-of-life improvements including adaptive equipment and educational support
- Staying informed about legitimate clinical trials through clinicaltrials.gov
Delaying proven orthopedic interventions while waiting for stem cell therapy approval can allow preventable complications to progress. Resources are better allocated to interventions with documented efficacy.
How to Evaluate Stem Cell Claims: A Family Protection Checklist
Red flags indicating potential exploitation:
- Clinics guaranteeing results
- Treatments requiring international travel
- Lack of published research
- Absence of institutional oversight
- High-pressure sales tactics
Green flags indicating legitimate research:
- FDA-registered trials
- Institutional review board approval
- Published protocols
- Transparent informed consent
- Research-funded or no-cost treatment
Families should verify clinical trial legitimacy through clinicaltrials.gov and ask specific questions: What is the FDA registration number? Where is the published safety data? What are the specific risks?
The Ethical Responsibility: Protecting Families While Advancing Science
Unicorn Biosciences maintains clear scope boundaries—orthopedic specialists treating musculoskeletal complications, not claiming to cure neurological CP. This transparent communication about what regenerative therapies can and cannot accomplish protects families from exploitation while providing access to legitimate solutions.
The commitment to FDA compliance and evidence-based practice reflects an ethical obligation to families navigating complex treatment landscapes. Supporting legitimate research while protecting vulnerable populations represents the responsible path forward.
Conclusion
The two-pathway framework remains essential for CP families in 2026: experimental neurological stem cell therapy has not yet achieved FDA approval, while proven orthopedic interventions are available now. The legitimate hope driving stem cell research deserves acknowledgment, but families need accurate information about current regulatory status.
Cerebral palsy’s musculoskeletal complications—contractures, joint deterioration, spasticity-related orthopedic issues—are treatable today with evidence-based care. Unicorn Biosciences offers FDA-compliant regenerative solutions within appropriate orthopedic scope, providing real options without the risks of stem cell tourism.
Families can access effective orthopedic care today while staying informed about legitimate research developments. Proven interventions should not be delayed in pursuit of experimental treatments that may be years from approval.
Take the Next Step: Expert Orthopedic Evaluation for CP Complications
Families seeking evaluation for musculoskeletal complications can schedule a consultation with Unicorn Biosciences to discuss FDA-compliant regenerative therapies. The personalized evaluation process includes imaging review, medical history assessment, and discussion of evidence-based treatment options.
Virtual and in-person consultations are available across multiple U.S. locations including Austin, Dallas, Houston, Fort Worth, San Antonio, El Paso, Boca Raton, and Manhattan. The commitment to transparency means honest assessment of what falls within orthopedic scope, with appropriate referrals when neurological interventions are being considered.
Contact Unicorn Biosciences at (737) 347-0446 to begin the conversation about proven solutions available today. Effective orthopedic care for CP complications does not require waiting for experimental treatments or risking unregulated overseas clinics.
Schedule Your Consultation Today!