Cellular Therapy for Ligament Tears: The Partial vs. Complete Tear Decision Framework
Cellular Therapy for Ligament Tears: The Partial vs. Complete Tear Decision Framework
When a patient receives a ligament tear diagnosis, confusion often follows. Should they pursue cellular therapy or proceed directly to surgery? The answer depends on several critical factors that many patients never learn to evaluate properly.
As of 2026, cellular therapy for ligament tears remains an evolving field. While the BEAR Implant has received FDA clearance, no stem cell products have been approved specifically for orthopedic conditions. This regulatory reality shapes the decision framework patients must navigate.
The path forward depends on tear classification, ligament type, anatomical factors, and evidence-based criteria. This article provides specific questions to ask physicians and identifies red flags indicating when surgery becomes necessary versus when cellular therapy offers a viable alternative.
While ACL tears dominate public discussion, MCL tears represent approximately 40% of knee ligament injuries and deserve equal consideration in any comprehensive treatment framework.
Understanding Ligament Anatomy: Why Some Heal Better Than Others
Ligaments possess inherently poor healing capacity. Only about 5% of ligament tissue consists of cells, with limited vascularization throughout the structure. This biological reality explains why some ligament injuries heal naturally while others require intervention.
The critical difference between ligaments lies in their blood supply. The MCL benefits from relatively superior healing capacity due to better vascularization. In contrast, the ACL suffers from poor blood supply, which explains why ACL tears rarely heal naturally while MCL tears often respond well to conservative treatment.
Blood supply delivers essential healing factors to injured tissue. Without adequate vascularization, the body cannot transport the growth factors, stem cells, and nutrients necessary for tissue repair. This fundamental principle determines cellular therapy candidacy for different ligament injuries.
Cellular therapies aim to overcome these natural healing limitations by delivering concentrated growth factors and stem cells directly to injury sites, essentially compensating for what the body cannot provide on its own.
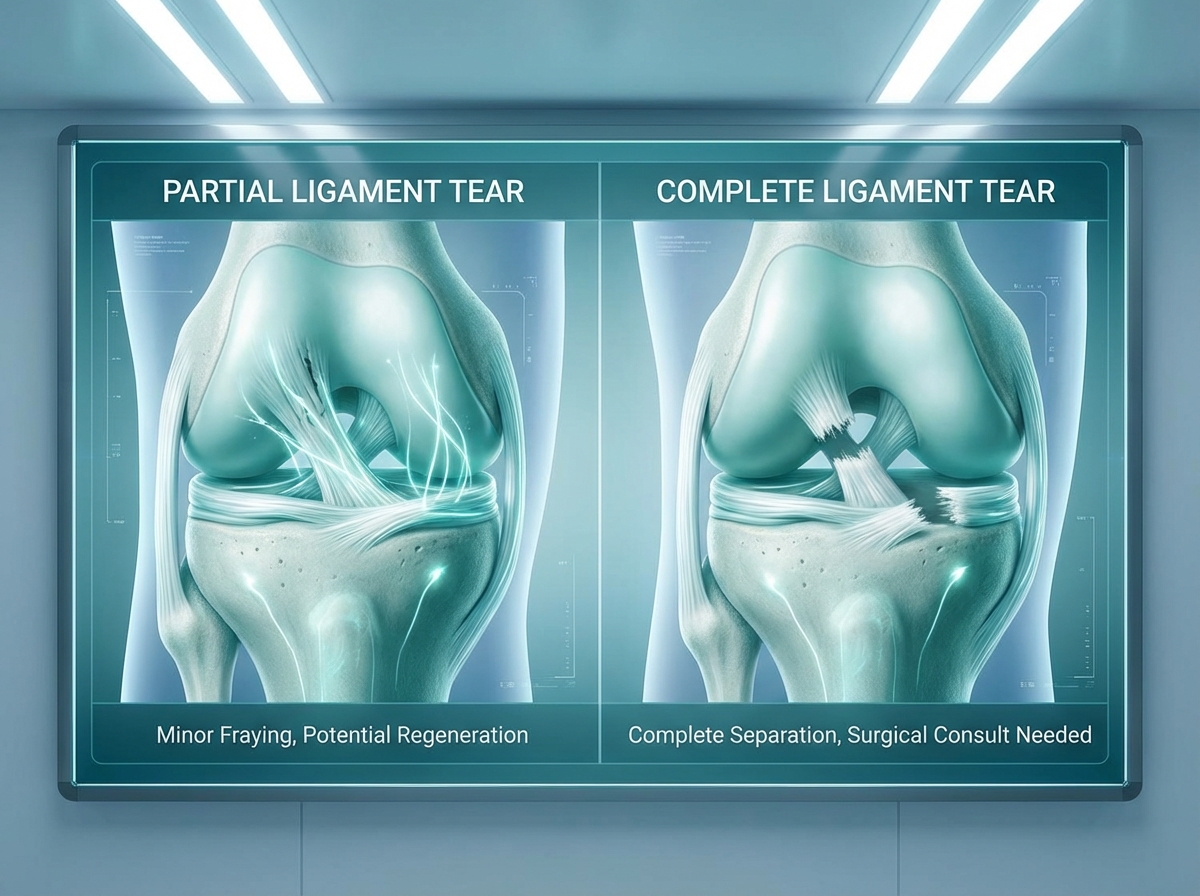
The Three-Tier Tear Classification System
Understanding tear classification is essential for treatment selection:
Partial Tears involve incomplete disruption of ligament fibers with an intact outer sheath. These represent the best candidates for cellular therapy because the remaining structure provides a scaffold for healing.
Complete Non-Retracted Tears are full-thickness tears where the ligament ends remain in proximity to each other. These patients may still be cellular therapy candidates depending on other factors.
Complete Retracted Tears occur when full-thickness tears cause significant separation between ligament ends. These injuries typically require surgical reconstruction because cellular therapy cannot bridge substantial gaps.
The “outer sheath” concept matters significantly. An intact sheath provides a scaffold for healing and keeps blood and cellular therapy contained at the injury site. MRI imaging is essential for accurate classification and treatment planning.
Current Cellular Therapy Options for Ligament Tears in 2026
Three main cellular therapy types dominate the current landscape:
- Mesenchymal Stem Cells (MSCs) derived from bone marrow or adipose tissue
- Platelet-Rich Plasma (PRP) concentrated from the patient’s own blood
- Bone Marrow Aspirate Concentrate (BMAC) providing both MSCs and growth factors
Autologous cells—those harvested from the patient’s own body—are preferred over allogeneic sources to avoid immune rejection and inflammatory responses.
The regulatory context remains critical: as of 2026, the FDA has not approved stem cell therapies specifically for orthopedic conditions. Only cord blood-derived hematopoietic stem cells for blood disorders carry FDA approval. Patients should understand this distinction when evaluating treatment options.
The BEAR Implant: Expanded 2025 FDA Clearance
The Bridge-Enhanced ACL Restoration (BEAR) Implant represents a significant advancement in ligament treatment. In March 2025, the FDA granted expanded 510(k) clearance for partial ACL tears and all ages, including children and adolescents.
The BEAR Implant works by providing a collagen scaffold saturated with the patient’s own blood, creating an environment conducive to ACL healing. Clinical trial data demonstrates non-inferior outcomes compared to ACL reconstruction at 2 years, with a 15% aggregate re-tear rate across trials.
More than 5,000 patients have received treatment with the BEAR Implant to date. Notably, the device shows superior hamstring muscle strength preservation compared to traditional ACL reconstruction—an important consideration for athletes.
Candidacy criteria include acute tears within weeks of injury, partial tears, and specific tear patterns. Patients should understand the distinction between FDA clearance (510(k)) and FDA approval, as these represent different regulatory pathways.
Mesenchymal Stem Cells and PRP: Evidence and Applications
Clinical studies show that approximately 75% of ACL tears treated with stem cells and PRP exhibited MRI evidence of healing. However, research remains limited, and long-term outcomes beyond 2 years continue under investigation.
A counterintuitive finding emerged from MCL research: low-dose MSCs (1×10⁶ cells) actually outperform high-dose treatments (4×10⁶) for MCL healing, producing increased failure strength and stiffness. Different doses uniquely affect cellular response and cytokine expression.
BMAC offers dual benefits by providing concentrated MSCs alongside growth factors. It can be used independently or as surgical augmentation. These therapies are typically used off-label without specific FDA approval for ligament tears.
Ligament-Specific Treatment Considerations
ACL Tears: Partial tears and complete non-retracted tears may be cellular therapy candidates. Complete retracted tears typically require surgical reconstruction.
MCL Tears: These injuries possess the best natural healing capacity among knee ligaments. Cellular therapy can potentially accelerate healing timelines.
LCL and PCL Tears: Less research exists for these ligaments, but similar principles apply based on tear classification and vascularization patterns.
Combination injuries involving multiple ligaments require individualized treatment protocols developed by experienced orthopedic specialists.
The Clinical Decision Framework: Surgery vs. Cellular Therapy
Criteria Favoring Cellular Therapy:
- Partial tears with intact outer sheath
- Complete non-retracted tears
- Acute injuries rather than chronic conditions
- Patient commitment to rehabilitation protocols
Red Flags Requiring Surgery:
- Complete retracted tears with significant gaps
- Chronic instability affecting daily activities
- Failed conservative treatment
- Multi-ligament injuries with structural instability
Patient factors including age, activity level, sport demands, and timeline for return to activity all influence treatment selection. If the knee gives way during daily activities—not just sports—surgery likely becomes necessary.
Some patients benefit from combination approaches where surgery is augmented with cellular therapy for enhanced healing outcomes.
Comparing Outcomes: What the Evidence Shows
ACL Reconstruction Surgery:
- 10-20% re-tear rate
- Only 65% of patients return to pre-injury performance levels
- 6-12 months recovery timeline
Cellular Therapy:
- Initial improvement in 4-6 weeks
- Full healing over several months
- Limited long-term data beyond 2 years
BEAR Implant:
- 15% re-tear rate in clinical trials
- Non-inferior patient-reported outcomes at 2 years
- Superior hamstring strength preservation
Long-term considerations matter: research suggests that a significant percentage of ACL-injured knees develop osteoarthritis over time. Studies show varying rates depending on factors such as meniscal injury and treatment approach. Research suggests cellular therapies may reduce this risk, though more investigation is needed.
Recovery Timeline and Rehabilitation Requirements
Cellular therapy patients typically see improvement within 4-6 weeks, with full healing occurring over several months. Surgical patients face 6-12 months of recovery with extended rehabilitation protocols.
Cellular therapy is not a shortcut. Committed rehabilitation remains essential regardless of treatment choice. Premature return to activity increases re-tear risk with any treatment method.
Cost Considerations and Insurance Coverage
Cellular therapies typically lack insurance coverage, costing several thousand dollars out-of-pocket. The BEAR Implant may have different coverage as an FDA-cleared device, though coverage varies by insurer.
Traditional ACL reconstruction generally receives insurance coverage with standard deductibles and co-pays. Patients should calculate total costs including procedure fees, rehabilitation, time off work, and potential revision surgery expenses.
Questions to Ask Your Physician
Patients should bring these questions to their consultation:
- What is the specific tear classification?
- Does the ligament tear have an intact outer sheath visible on MRI?
- Is the patient a candidate for the BEAR Implant?
- What is the physician’s experience with cellular therapy for ligament tears?
- What are re-tear rates for similar patients?
- If cellular therapy fails, what surgical options remain?
- What rehabilitation protocol will be followed?
- What will this cost out-of-pocket?
- Is the physician using FDA-cleared devices or off-label therapies?
- What red flags indicate surgery is necessary instead?
Red Flags That Indicate Surgery Is Necessary
Surgery becomes necessary when patients present with:
- Complete retracted ACL tears with significant gaps on MRI
- Chronic instability affecting daily activities
- Multi-ligament injuries with structural instability
- Failed conservative treatment or cellular therapy
- Significant meniscus tears requiring surgical repair
- Inability to commit to extensive rehabilitation
The Future of Cellular Therapy for Ligament Tears
A $140 million funding round announced in January 2026 is supporting late-stage clinical development of cell-based therapies for inflammatory and degenerative conditions. Currently, 224 clinical trials globally are examining stem cell therapies for osteoarthritis and related conditions.
As evidence accumulates, FDA approval pathways may become clearer for specific cellular therapy products. The field continues evolving with new techniques, dosing protocols, and combination approaches emerging regularly.
Conclusion
The partial versus complete tear distinction serves as the primary decision point for cellular therapy candidacy. Ligament type matters significantly—the MCL’s superior healing capacity versus the ACL’s poor vascularization affects treatment selection.
As of 2026, only the BEAR Implant has FDA clearance for ACL repair, while stem cell therapies remain off-label. Accurate tear classification via MRI and consultation with experienced orthopedic specialists remain essential.
Both cellular therapy and surgery require committed rehabilitation. Neither offers an easy solution. Patients should use this decision framework and the physician questions provided to make informed choices aligned with their specific injury, goals, and circumstances.
Schedule Your Consultation
Unicorn Bioscience offers comprehensive evaluations for patients considering cellular therapy for ligament tears. With virtual and in-person consultations available across eight locations in Texas, Florida, and New York, patients can access expert guidance regardless of location.
The clinic’s precision-guided cellular therapy utilizes advanced ultrasound and X-ray imaging for accurate delivery to targeted treatment areas. Personalized treatment plans address individual factors including inflammation levels, age, injury type, and health goals.
Qualified candidates may receive same-day treatment following consultation. To schedule an evaluation, call (737) 347-0446 or visit unicornbioscience.com. Patients should bring their MRI results and prepared questions to ensure a comprehensive evaluation of their treatment options.
Schedule Your Consultation Today!