Alternatives to Knee Replacement Surgery: The Multi-Modal Treatment Protocol That Achieves 90% Surgical Avoidance
Alternatives to Knee Replacement Surgery: The Multi-Modal Treatment Protocol That Achieves 90% Surgical Avoidance
The statistics are striking: studies suggest that 80% of patients told they need total knee replacement may not actually require surgery. This finding represents a fundamental shift in how orthopedic medicine approaches knee osteoarthritis, moving away from the traditional surgical pathway toward comprehensive, multi-modal cellular therapy protocols that address the root causes of joint deterioration.
With over 700,000 knee replacement surgeries performed annually in the United States at an average cost ranging from $20,000 to $35,000 per procedure, the stakes extend far beyond individual patient outcomes. When approximately 10-20% of knee replacement patients report dissatisfaction with their results and approximately 20% experience chronic pain post-surgery, the medical community has been compelled to develop more effective alternatives.
The emergence of precision-guided multi-modal cellular therapy protocols has changed the landscape dramatically. Clinics specializing in regenerative medicine now report that more than 90% of stem cell patients have not progressed to knee replacement surgery—a success rate that demands attention from anyone facing this major surgical decision.
Why Single-Treatment Approaches Are Becoming Obsolete
Traditional approaches to knee osteoarthritis treatment have relied on isolated therapies: PRP alone, stem cells alone, or hyaluronic acid injections as standalone interventions. While each modality demonstrates measurable benefits, the ceiling effect of single-treatment approaches has become increasingly apparent.
The MILES study published in Nature found that mesenchymal stem cells performed equally to corticosteroid injections for knee osteoarthritis pain relief up to one year. While this demonstrates safety and efficacy, it also highlights that isolated cellular therapy may not unlock the full regenerative potential these treatments offer.
Knee osteoarthritis is inherently multi-factorial. The condition involves simultaneous inflammation, cartilage degradation, bone marrow lesions, and synovial dysfunction. Addressing only one mechanism while leaving others untreated limits therapeutic outcomes. The solution lies in strategic combination protocols that create synergistic effects—where the combined impact exceeds what any single treatment achieves independently.
The Multi-Modal Cellular Therapy Protocol: A Comprehensive Framework
A true multi-modal protocol differs fundamentally from simply receiving multiple treatments sequentially. It involves strategic sequencing and timing of different regenerative modalities, each selected to address specific pathological mechanisms while enhancing the effectiveness of companion therapies.
The core components typically include:
- Platelet-Rich Plasma (PRP) for anti-inflammatory foundation
- Mesenchymal Stem Cells (MSCs) for tissue regeneration
- Exosomes for enhanced cellular communication
- Hyaluronic Acid for joint lubrication and scaffold support
- Targeted biologics customized to individual patient profiles
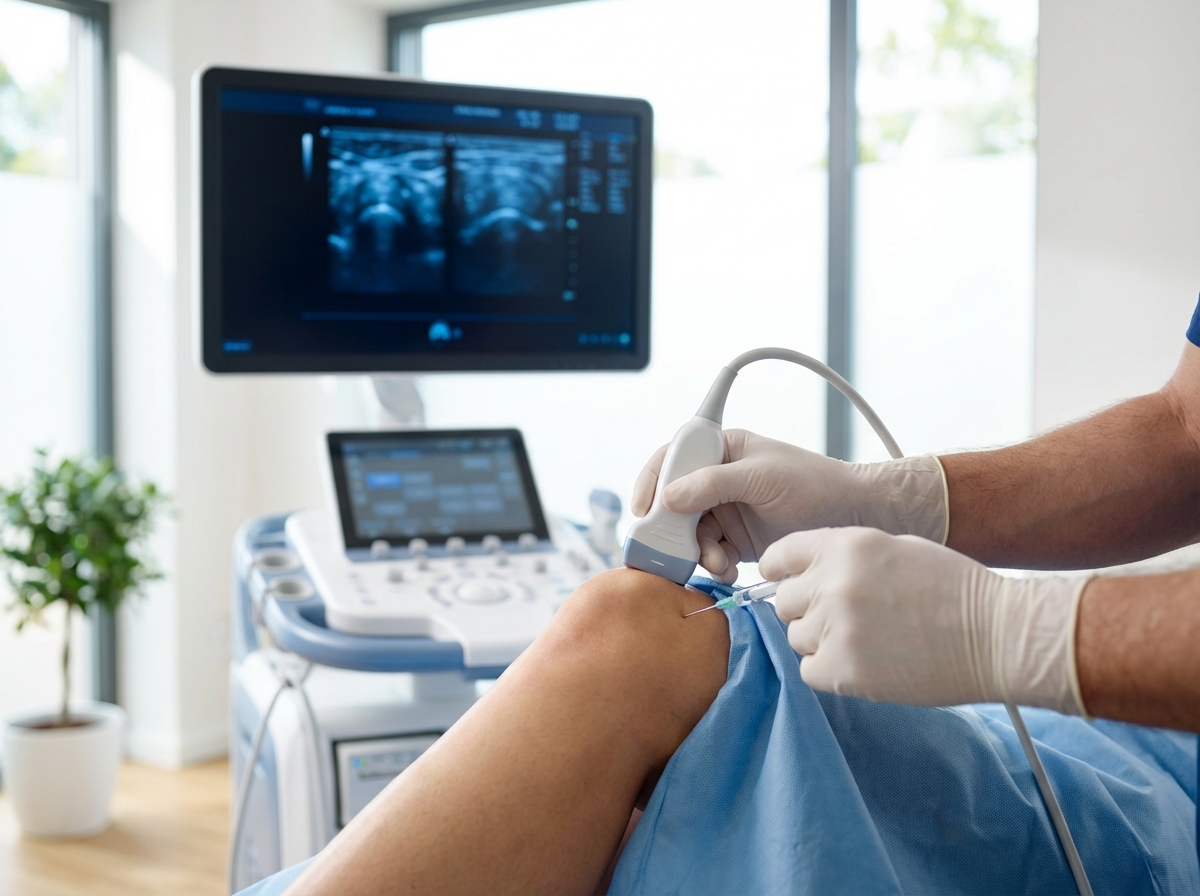
Critical to success is precision-guided delivery. All injections should be administered using advanced imaging guidance, including ultrasound and fluoroscopic technology, ensuring accurate delivery of therapeutic agents to targeted treatment areas.
Component 1: Platelet-Rich Plasma (PRP) as the Anti-Inflammatory Foundation
PRP injections demonstrate superior pain relief and functional improvement compared to hyaluronic acid and corticosteroids, particularly for patients with Kellgren-Lawrence grades I-III (mild to moderate osteoarthritis). This makes PRP the ideal foundation for comprehensive treatment protocols.
Within the multi-modal framework, PRP serves to reduce inflammation and prepare the joint environment for subsequent regenerative therapies. The concentrated platelets release growth factors that initiate healing cascades while modulating the inflammatory response that perpetuates cartilage breakdown.
PRP concentration variables must be tailored to individual inflammation levels. While PRP alone shows 60-70% improvement rates, its true value emerges when combined with cellular therapies—creating an optimized environment for stem cell survival and differentiation.
Component 2: Mesenchymal Stem Cells (MSCs) for Tissue Regeneration
Mesenchymal stem cell therapy demonstrates 60-80% success rates for mild to moderate knee osteoarthritis, with patients experiencing 58-68% pain reduction and 67-83% functional improvement at one to two years. These outcomes reflect MSCs’ dual mechanism: immunomodulation and tissue regeneration through paracrine signaling.
A comprehensive review of 15 randomized controlled trials published in Nature showed net positive effects on pain and function from MSC therapy. Importantly, stem cells target bone marrow lesions—increasingly recognized as the actual source of pain in many knee osteoarthritis cases, rather than cartilage loss alone.
Bone marrow aspiration concentrate (BMAC) provides autologous MSCs with minimal processing, maintaining the natural cellular environment. When combined with PRP, MSC survival rates and differentiation capacity improve significantly, explaining why integrated protocols outperform isolated stem cell injections.
Component 3: Exosomes for Enhanced Cellular Communication
Exosomes are extracellular vesicles that facilitate cellular communication and tissue repair. Within multi-modal protocols, they amplify regenerative signals from stem cells and PRP, enhancing the therapeutic cascade throughout the joint environment.
Emerging evidence supports exosomes’ role in cartilage protection and anti-inflammatory effects. These nanoscale messengers carry proteins, lipids, and genetic material that instruct recipient cells to initiate repair processes.
As of 2026, the FDA has not approved stem cell, PRP, or exosome products specifically for orthopedic conditions. However, substantial clinical evidence supports safety and efficacy when administered by qualified providers within FDA regulatory frameworks.
Component 4: Hyaluronic Acid for Joint Lubrication and Scaffold Support
Rather than functioning as a standalone treatment, hyaluronic acid serves a strategic role within comprehensive protocols. Viscosupplementation provides mechanical cushioning while biologics perform their regenerative work, reducing friction-induced damage during the healing period.
Additionally, hyaluronic acid functions as a scaffold for cellular therapies, helping retain stem cells and growth factors within the treatment area. Molecular weight considerations affect treatment duration, with higher molecular weight formulations providing longer-lasting lubrication effects.
Precision Patient Profiling: Matching Protocols to Individual Phenotypes
Osteoarthritis presents in distinct phenotypes and endotypes requiring personalized treatment selection. A comprehensive evaluation considers:
- Inflammation levels (assessed through imaging and biomarkers)
- Patient age and activity level
- Kellgren-Lawrence grade (I-IV severity classification)
- Presence and location of bone marrow lesions
- Current medications and comorbidities
Advanced imaging using MRI, ultrasound, and X-ray guides protocol customization. Inflammatory-dominant osteoarthritis requires different protocol emphasis than mechanical-dominant presentations. This precision medicine approach, targeting dysregulated mechanisms rather than applying one-size-fits-all treatment, represents the current direction of osteoarthritis care.
Clinical Outcomes: The 90% Surgical Avoidance Success Rate
The evidence supporting multi-modal protocols continues to strengthen. Research published in the Journal of Orthopaedic Experience & Innovation found that 84% of total knee replacement candidates avoided surgery at two years following comprehensive intervention. Pain decreased by 42%, and function improved by 39% at six months.
Clinics implementing comprehensive multi-modal cellular therapy protocols report that more than 90% of stem cell patients have not progressed to knee replacement surgery. This compares favorably to single-modality approaches showing 60-70% success rates.
Realistic expectations remain essential. Multi-modal protocols demonstrate greatest effectiveness for Kellgren-Lawrence grades I-III (mild to moderate osteoarthritis). Patients with severe bone-on-bone deformity may still require surgical intervention, though even advanced cases often benefit from reduced pain and improved function.
Emerging Innovations Enhancing Multi-Modal Protocols
Several breakthrough developments are expanding treatment possibilities:
Genicular Artery Embolization (GAE) targets inflammation through reduced blood flow to abnormal vessels in the knee. This minimally invasive procedure demonstrates 99.7% technical success rates with pain reduction of 34-39 points on the Visual Analog Scale.
Anti-aging research from Stanford Medicine (November 2025) revealed that blocking the 15-PGDH protein regenerates knee cartilage in mice and human tissue samples. This breakthrough suggests actual cartilage regeneration—rather than symptom management—may become achievable, potentially eliminating the need for replacement in many patients.
Gene therapy advances continue progressing, with the FDA granting Regenerative Medicine Advanced Therapy designation to GNSC-001, a first-in-class gene therapy blocking IL-1. Phase IIb/III trials are expected to initiate in 2026.
Who Is—and Isn’t—a Candidate for Multi-Modal Cellular Therapy
Ideal candidates include:
- Patients with Kellgren-Lawrence grades I-III osteoarthritis
- Those with identifiable bone marrow lesions
- Individuals with inflammatory osteoarthritis phenotypes
- Surgery-averse patients seeking alternatives
- Younger patients facing the reality of knee replacement implants that may require revision
Contraindications include:
- Severe bone-on-bone deformity (grade IV)
- Significant joint malalignment requiring surgical correction
- Unrealistic expectations for complete restoration
Research indicates that approximately one-third of knee replacement patients have arthritis symptoms that may not be severe enough to warrant surgery—making them prime candidates for comprehensive cellular therapy protocols.
Comparing Multi-Modal Protocols to Knee Replacement Surgery
The comparison reveals significant advantages for appropriate candidates:
| Factor | Multi-Modal Protocol | Knee Replacement |
|---|---|---|
| Recovery Time | Same-day to minimal | 3-6 months |
| Dissatisfaction Rate | Low | 10-20% |
| Chronic Pain Risk | Minimal | Approximately 20% |
| Mortality Risk | Negligible | 0.35% within 90 days |
| Revision Rate | Maintenance protocols | Variable by age |
| Reversibility | Preserves surgical options | Irreversible |
Multi-modal protocols preserve future surgical options if needed, while surgery represents an irreversible decision with a finite implant lifespan.
What to Expect: The Multi-Modal Treatment Journey
The process begins with comprehensive evaluation including advanced imaging, inflammation assessment, and phenotype identification. Based on these findings, a personalized protocol is developed addressing the specific pathological mechanisms present in each patient’s condition.
Precision-guided injection procedures utilize ultrasound and X-ray guidance to ensure accurate delivery. Qualified candidates may receive treatment on the same day as their consultation, streamlining the patient experience.
The typical treatment timeline includes an initial anti-inflammatory phase, followed by regenerative treatment, with maintenance protocols as needed. Patients can expect initial relief within weeks, with continued improvement over 3-12 months as cellular regeneration progresses.
Navigating the Regulatory Landscape and Choosing Qualified Providers
With 224 clinical trials globally investigating stem cell therapies for osteoarthritis—including a $140 million Phase III trial announced in January 2026—the evidence base continues expanding rapidly.
Patients should seek board-certified physicians with experience in precision-guided injections and multi-modal approaches. U.S.-based treatment within FDA regulatory frameworks provides important safeguards compared to medical tourism options.
Providers offering comprehensive evaluation rather than one-size-fits-all protocols demonstrate the personalized approach necessary for optimal outcomes. Patients should be cautious of clinics making “guaranteed” claims not supported by evidence.
Conclusion
The paradigm shift from single-treatment approaches to precision-guided multi-modal cellular therapy protocols represents a fundamental advancement in knee osteoarthritis care. With 90% surgical avoidance success rates when comprehensive protocols are properly applied, patients facing knee replacement recommendations deserve to understand their alternatives.
For the 80% of patients who may benefit from non-surgical treatment, multi-modal cellular therapy offers an evidence-based, personalized medicine approach addressing the multi-factorial nature of knee osteoarthritis. While not appropriate for all patients, these protocols represent the current standard for maximizing non-surgical outcomes.
Take the Next Step Toward Avoiding Knee Replacement Surgery
Patients considering their options should seek comprehensive evaluation from qualified regenerative medicine providers. Unicorn Bioscience offers precision-guided multi-modal protocols combining PRP, stem cells, exosomes, and hyaluronic acid tailored to individual patient profiles.
With same-day treatment availability for qualified candidates across eight locations in Texas, Florida, and New York, both virtual and in-person consultations provide accessible pathways to evaluation. All treatments are administered in the United States within FDA regulatory frameworks.
To begin the journey toward surgical avoidance with comprehensive imaging-guided evaluation and personalized protocol design, contact Unicorn Bioscience at (737) 347-0446 or visit unicornbioscience.com.
Schedule Your Consultation Today!